Medical Device Certification
Your Business, our focus
Eurofins Product Testing Italy is a Notified Body No. 0477 for CE certification in accordance with Regulation (EU) 2017/745 and Certification Body no. 00054 for quality system verification according to the ISO 13485 scheme
Medical Device Certification - MDR (EU) 2017/745
Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017 on Medical Devices (hereinafter "MDR"), which repeals Directive 90/385/EEC (AIMDD) and Directive 93/42/EEC (hereinafter "MDD"), entered into force on 25 May 2017 and applies from 26 May 2021.
Eurofins Product Testing Italy Srl, since 22/07/2021, is Notified Body to operate according to the provisions of Regulation (EU) 2017/745 (MDR) for CE certification of Active and Non-Active Medical Devices of Class I (Is, Im, IR), IIa, IIb, and III.
The list of devices for which Eurofins Product Testing Italy operates is available on the NANDO system.
What is a Medical Device (Art.2)
‘Medical Device’ means any instrument, apparatus, appliance, software, implant, reagent, material or other article intended by the manufacturer to be used, alone or in combination, for human beings for one or more of the following specific medical purposes:
- diagnosis, prevention, monitoring, prediction, prognosis, treatment, or alleviation of disease,
- diagnosis, monitoring, treatment, alleviation of, or compensation for, an injury or disability,
- investigation, replacement or modification of the anatomy or of a physiological or pathological process or state,
- providing information by means of in vitro examination of specimens derived from the human body, including organ, blood and tissue donations, and which does not achieve its principal intended action by pharmacological, immunological or metabolic means, in or on the human body, but which may be assisted in its function by such means.
The following products are also considered Medical Devices:
- devices for the control or support of conception,
- products specifically intended for the cleaning, disinfection or sterilization of devices as referred to in Article 1(4) and of those referred to in the first paragraph of this point.
The MDR also governs:
- Medical Devices accessories
- Groups of product without an intended medical purpose listed in Annex XVI
- Device which incorporates, as an integral part, a substance which, if used separately, would be considered to be a medicinal product … and that has an action ancillary to that of the device.
- Device which is intended to administer a medicinal product
- Devices that are also machinery
Manufacturer Obligations
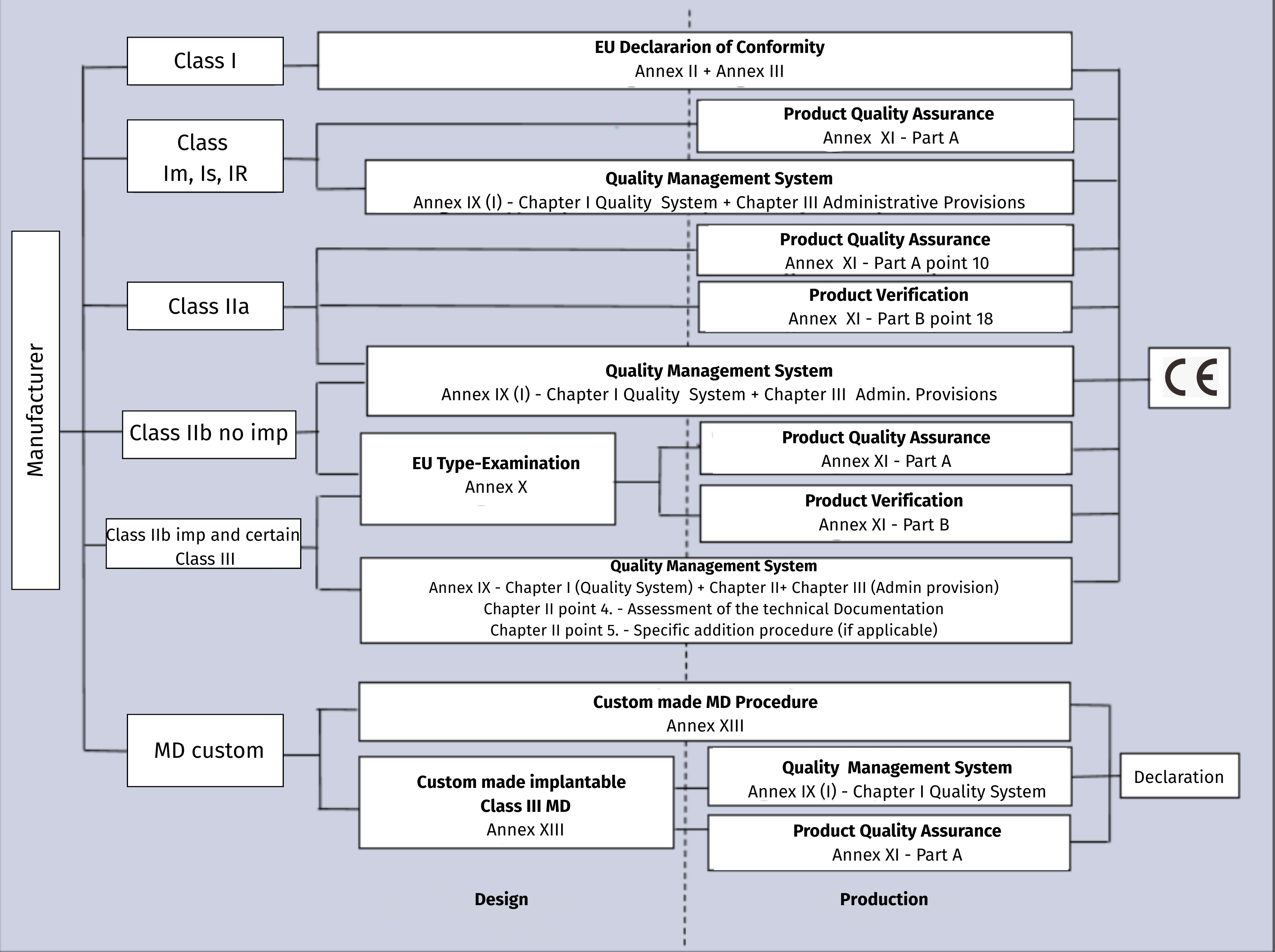
CE marking and, consequently, the placing on the market of Medical Devices in the countries of the European Community, is regulated by Regulation (EU) 2017/745. Medical devices are classified according to the risk posed by their nature and are divided into classes:
> Class I Low/medium risk devices
> Class IIa Medium risk devices
> Class IIb Medium/high risk devices
> Class III High risk devices
Eurofins Product Testing Italy is a Notified Body No. 0477 No. 0477 for CE certification in accordance with Regulation (EU) 2017/745 for Active and Non-Active Medical Devices of Class III, IIb, IIa and Class I (ie Im: Measuring devices, Is: Sterile devices and Ir: Reusable devices).
Certification Path
Management System Certification - ISO 13485
Eurofins Product Testing Italy also operates as an accredited Certification Body for quality system verification according to the ISO 13485 scheme (accreditation no. 00054 Management System Certification - ex SGQ no. 133A).
Certification according to ISO 13485 attests to the reliability of the quality management system adopted by companies in the medical sector. Below you will find the complete list of accredited Technical Areas:
- 1.1 NON-ACTIVE MEDICAL DEVICES
1.1.A General non-active, non-implantable medical devices
1.1.B Non-active implants
1.1.C Wound care devices
1.1.D Non-active dental devices and accessories
1.1.E Non-active medical devices other than those specified - 1.2 ACTIVE MEDICAL DEVICES
1.2.A General active medical devices
1.2.C Monitoring devices
1.2.D Devices for radio-therapy and thermo-therapy - 1.5 STERILIZATION METHODS FOR MEDICAL DEVICES
1.5.A Ethylene oxide gas (EOG) sterilization
1.5.B Moist heat
1.5.C Aseptic processing
1.5.D Radiation sterilization (e.g.: gamma rays, electron beam) - 1.6 DEVICES CONTAINING/USING SPECIFIC SUBSTANCES/TECHNOLOGIES
1.6.A Medical devices incorporating medicinal substances
1.6.B Medical devices utilizing tissues of animal origin
1.6.F Medical devices utilizing biological active coating and/or materials entirely or mainly - 1.7 PARTS AND SERVICES
1.7.E Distribution Services
The list of accredited schemes UNI CEI EN ISO/IEC 17021-1 by ACCREDIA can be consulted on the website www.accredia.it, accreditation no. 00054 Management System Certification (formerly QMS no. 133A)